Digitization in Dentistry and Dental Technology

For most people, a visit to the dentist or orthodontist is joyless but necessary. This may be partly because many dental treatments, such as taking an impression of the jaw and teeth, is generally experienced as unpleasant. New technologies are set to make such treatments much more pleasant in the future – for example, by using an intraoral scanner to take an impression of the jaw. But this is just a tiny part of the digital revolution in dentistry. Given that dentistry is such a high-tech field, the digital revolution, along with all of the associated innovations it brings, is being welcomed with open arms.
Digital Treatment Methods
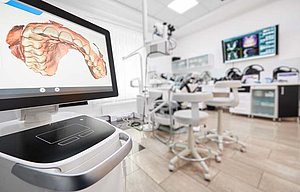
Some treatment steps are already being supported with digital technologies, or have been completely replaced. For example, the use of sanding machines to provide some dental services is on the agenda. The latest technologies for orthodontic or dental 3-D diagnostics can not only be used for endodontic treatment and to plan operations – they also make it possible to make implants unique to the patient for immediate use. Digital patient information systems have also been developed for the post-treatment period. These can analyze and display the expected influence of the therapy options on facial and dental esthetics. These technologies only represent a fraction of the digital support options available in dentistry. In addition to the technologies already in use, other innovations are indicating the direction in which the dental industry is likely to move.
A Glimpse into the Future
The latest developments are already pointing the way toward potential future innovations in technology. In China, robots with an integrated medical expert system are already being used. The information that the robots collect and analyze during the treatment is used to support the medical staff. Another promising step would be a global information system. With a high level of digitization, it may be possible to implement a globally consistent system that learns and shares information in real time. This may enable findings about complications during an operation in the US to be shared directly with medical staff in Germany, who can then take this information into account the next time they have to perform surgery. In this way, potential complications could be minimized, or even prevented, before the surgery even takes place. It is also conceivable that artificial intelligence could be used to evaluate health-related data in order to speed up the analysis of treatment failures and complications.
In order to be able to use these technologies to their fullest extent, however, the ethical and legal aspects first need to be clarified. For example, it must be clearly defined who is liable if a machine makes the wrong decision, or if a patient is harmed as a result of a robot malfunction. It remains to be seen, therefore, which other developments will arise for dentistry and the dental technology field.
Advantages of Digitization
Digitization and the associated advances in medicine have plenty of positive aspects. In comparison to standard treatments, a digital workflow may be able to reduce the number of individual steps involved. By streamlining the process in this way, the doctors not only save time – it also reduces stress and error susceptibility. Another major advantage is the networking of medical devices. With such a network in place, devices can become intelligent and active elements within a modern healthcare system and optimize workflows and processes. The data collected then provide a comprehensive view of the patient. This results in more efficient treatment, higher patient safety and improved consulting and treatment options between doctors.
Simple digitization strategies can also improve efficiency in doctors’ offices – such as through paperless administration, for example. This can improve process security, reduce costs and ensure a more cost-effective business model. Furthermore, dental practices can stand out from the competition with their digital services and position themselves as more innovative.
Disadvantages of Digitization
In addition to a whole range of advantages, negative aspects have also been observed in the digitization process. Purchasing and updating digital devices is associated with high cost burdens for the end consumer of the devices, including dentists and orthodontists. For smaller practices, it remains to be seen whether these costs will pay off over time. Many devices also require a high level of expertise in order to operate them properly; for example, in order to intervene quickly if there are complications. Another negative aspect is the lack of human touch with these treatments. Many patients show a great degree of trust in their doctors – provided they are human. Robots and machines, however, face a higher level of skepticism.
The Role of Translation

Where medical devices are intended for global use, expert translations play an essential role. Professional translations are vital for both the manufacture of medical devices and in their sale and operation. mt-g can translate your advertising and information materials, but also handbooks and operating manuals for you. Only by doing so can your customers and interested parties receive the relevant information that is clearly written and easy to understand. This also ensures that the experts can then operate the products correctly and provide the perfect treatment.
However, the software included with the products also has to be localized for the specific target group. mt-g only works with specialist translators for this. They take into account the limited space available for dialog boxes, buttons and menus and have the necessary experience with software translation. In this way, despite the different lengths of the translated texts, the ideal results can be obtained to support the graphics of the user interface in the best possible way.
Additional language services are also essential once the medical product has been licensed. Any revision or development of a product requires the technical documentation to be updated, which is subject to the requirements of the EU Medical Device Regulation (MDR) or the In Vitro Diagnostic Regulation (IVDR). The mt-g Dental Team is not only happy to take care of the translation into the languages you require but also supports customers throughout the entire process.





